The Future of Personalised Medicine: AI and Your DNA

Healthcare is going to change a lot in the near future. AI will look at your DNA and suggest treatments that are as different from each other as your fingerprint. This mix of AI and genomics is making medicine go from a one-size-fits-all approach to one that is made just for you.
The Basics of History
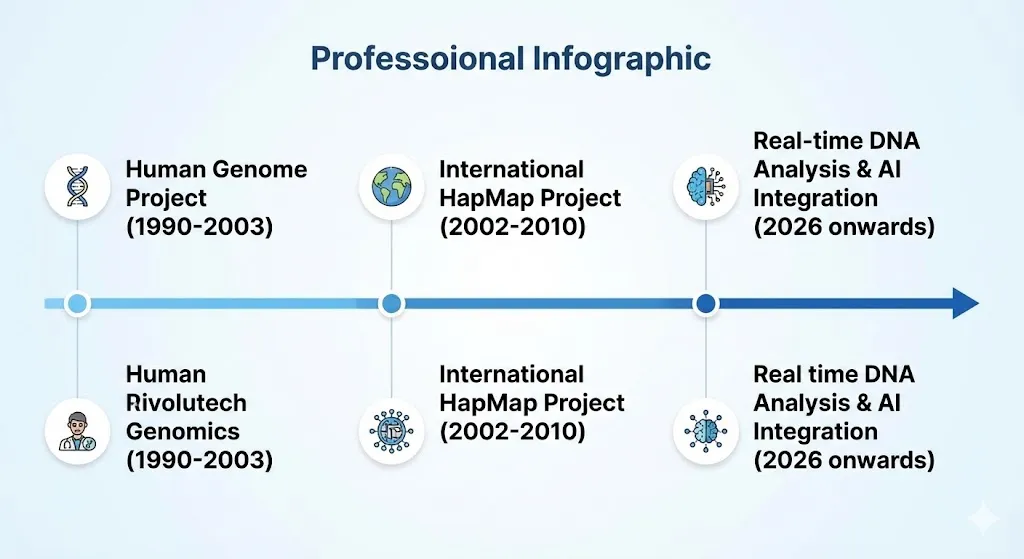
DNA sequencing became faster and easier to use in the 1990s, which helped personalised medicine become very popular. The Human Genome Project (1990–2003) mapped more than three billion base pairs, and the International HapMap Project (2002–2010) found genetic differences that were linked to diseases. These projects made it possible to make treatments that work for each person’s genes. We don’t use treatments that work for everyone anymore. We now use treatments that are based on each person’s genes. People first heard about this idea in 1999, when pharmacogenomics was becoming more popular. One of the first groups to link gene variations to specific diseases was the Single Nucleotide Polymorphisms Consortium. These projects taught us how the genome works. This helped us move from just treating symptoms to using methods that stop problems before they happen.
What’s going on now and what’s new?
Between 2025 and 2035, AI is expected to become more popular in the precision medicine market [web:2]. AI now uses large genomic datasets, clinical records, lifestyle factors, and environmental data to give us useful information. Pharmacogenomics is the study of how drugs work with different types of omics data. AI is very good at this. This is because it can find small interactions between drugs and genes. AI finds complicated variants for targeted therapies in oncology, which makes it easier to figure out what’s wrong and how bad it is. The market for AI in precision medicine is growing by 25.54% every year and will be worth $33.45 billion by 2035. In 2024, the bigger personalised medicine market was worth $567.10 billion. By 2033, it should be worth $1,196.18 billion, with a CAGR of 8.80%. The cost of sequencing is going down, and new genomic therapies are being made, which is why this growth is happening.

The AI market for precision medicine is expected to grow from 2025 to 2035 [web:2]. Asia-Pacific is growing the fastest, at a rate of 11.4% every year. People are living longer, and more money is going into China and India.
How AI Uses Your genes
AI algorithms look for changes, patterns in gene expression, and epigenetic markers in your genome, which has about 20,000 genes and billions of base pairs. DeepMind’s AlphaFold and other deep learning models use sequences to figure out what proteins look like. This helps us learn more about how drugs work and makes it easier to find new ways to help people. We use recurrent networks to look at sequences and convolutional neural networks to find variants. This turns raw FASTQ files into profiles that doctors can use. AI looks at your genomic data and compares it to big databases when you use it. After that, it uses generative models to show how the treatments could work. This lets you change your medications in real time based on pharmacogenomic feedback to cut down on side effects.
Real-World Impact and Case Studies
23andMe uses AI to look at millions of user genomes to find signs of Type 2 diabetes and Parkinson’s disease. It shares information without names to speed up the process of finding new drugs. Their platform is an example of consumer-driven precision medicine because it helps people make changes to their lives before they have to. IBM Watson for Oncology, which was made with Memorial Sloan Kettering, looks at a patient’s genetics and medical history to suggest treatments that are in line with tumour boards in 93% of breast cancer cases. More people have answered, and they have lived longer without getting cancer. Grail and Illumina’s AI-powered liquid biopsies look for circulating tumour DNA, which helps them find cancers before they show any signs with a high level of accuracy. In one study, it found a number of cancers early on, which led to treatments that saved lives. Watson and the Mayo Clinic work together to make better personalised cancer treatment plans than the ones that are usually used. They use genetic and historical information to do this. DeepCare at UCSF uses electronic health records (EHRs) and genomics to make predictions about the health of 100,000 patients and find new risks. These examples show how AI can really help by making it less likely that people will have to try a lot of different treatments before they find one that works.
Market Data and Predictions
The expected value (in USD Bn) for the market segments in 2024/2025 and 2030/2035: AI Precision Medicine: 3.44 (2025), 33.45 (2035), 25.54; Personalised Medicine Overall: 567.10 (2024), 1,196.18 (2033), 8.80; Therapeutics Subsegment: N/A; Fastest at 10.2% to 2030; 10.2; Projections show that there will be 28 new personalised treatments in 2023 alone, and AI will cut drug discovery time from years to months. Genomic AI platforms will be able to change dosages on the fly by 2026.
Problems with putting it into action
Even though it looks good, data privacy is a big problem. Genomic data is very private, so it needs to be very well protected even if it gets out. AI can’t be fully integrated because electronic health records (EHRs) and genomic databases don’t work well together. There are rules that make it take longer for AI tools to get approved. Clinicians also aren’t getting the training they need to keep up with how smart algorithms are becoming. Equity raises moral questions because it pushes groups that aren’t represented enough to the edges. Things could get even worse because of the high cost of computing and the biassed training data.
New ideas and chances
AI opens up new possibilities in predictive analytics, such as using genomic signals to predict how diseases will get worse. New technologies, like pharmacogenomics, which make medicines that heal themselves, can sometimes help reduce side effects. Partnerships like Google with Cancer Genome Atlas help researchers find new targets for treatment. Genomics is easier to understand with tools like 23andMe, which also encourage people to take care of themselves. Edge computing will make it possible for wearables to do genomic analysis in real time.
What to Expect in the Future
By the year 2030, everyone will have their genome sequenced at birth, and AI companions will watch over their health for the rest of their lives. Multimodal AI will use genomics, wearables, and lifestyle data to make predictions that take all of these things into account. Quantum-enhanced AI could quickly simulate entire proteomes, which would allow for the creation of custom biologics. There are a lot of issues with climate-adaptive therapies and global health equity, but there are also a lot of opportunities. Personalised medicine will be the most popular, and AI triage will cut wait times by 60%.
The Best Ways to Adopt
Federated Learning: lets you teach AI without putting sensitive genomes in one place.
Interdisciplinary Teams: Work with geneticists and AI experts to make sure your models are right.
Think about the patient when you design: Use AI that can explain itself to make suggestions clearer. This will make people more likely to trust you.
As part of your moral framework: use bias audits and rules that follow the GDPR.
Pilot Scalability: Start with a small group of people with cancer and then add more. These steps make sure that the integration stays in place.
FAQ
What kind of medicine is made just for you?
AI and genomics are used in personalised medicine to make sure that when people get sick, their genes, environment, and way of life are all taken into account. It starts with broad actions and then moves on to more specific ones, which makes things better.
How does AI analyse my DNA?
AI looks at sequencing data, finds differences, and then uses models like AlphaFold to guess how they will act. It puts together data from many omics to make a complete picture.
How do people use AI in the real world?
23andMe tells you how likely you are to get sick, and Watson for Oncology suggests cancer treatments that 93% of doctors agree on. Grail’s biopsies help find things early on.
What problems does it have to deal with?
There are still problems with privacy, data silos, delays in following the rules, and unfair access. Federated learning and rules of ethics are two ways to solve the problem.
When will it be normal?
By 2030, the market will be bigger and the technology will be better. The $33 billion precision sector will be run by AI.