Drug Discovery at Warp Speed: How AI is Designing New Medicines
The pharmaceutical industry is about to go through a big change. AI is speeding up drug discovery from a long, slow process to a quick, accurate sprint. AI saves time, money, and the need for new treatments that seemed impossible before by using machine learning, generative models, and huge biomedical datasets.
Change as time goes on
Some things that happened by chance led to the discovery of drugs. For example, the moldy petri dish that led to the discovery of penicillin in 1928. It has changed over the years, going from high-throughput screening in the 1990s to tools that run on computers in the 2010s. Around 2015, we began to see AI integration. Atomwise and other early adopters used convolutional neural networks for virtual screening. This was a big change from trying things out with brute force to using predictive intelligence.
The following infographic illustrates this historical evolution, highlighting key milestones from early discoveries to the integration of AI.

Quantitative structure-activity relationship (QSAR) models in the 1960s laid the groundwork for what we know now. But things didn’t really get better until after 2020, when GPUs and cloud infrastructure made computers a lot more powerful. AlphaFold was released in 2020 and solved the problem of protein folding in days instead of years. This was a turning point that led to more than 200 million predictions of structures by 2025.
The key technologies that enable AI-driven discovery
Variational autoencoders and diffusion models are two examples of generative AI models that are leading the way in this field. They learn how to make new molecules from chemical libraries that contain billions of compounds. Graph neural networks help us understand how proteins and ligands work together. We can now predict how well things will bind together with 50% more accuracy than before.
This diagram visualizes the stack of AI technologies that power modern drug discovery, from data to models.
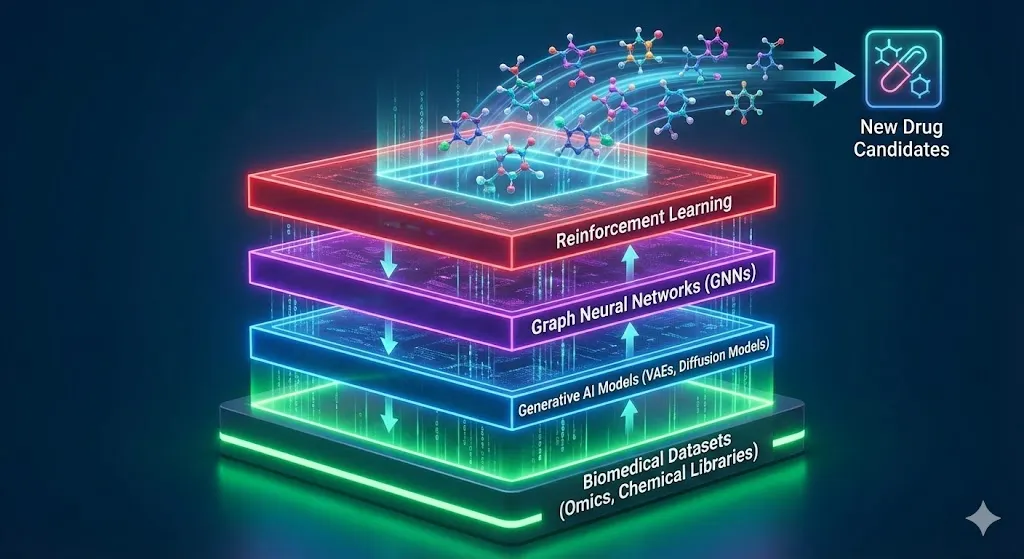
Reinforcement learning improves lead compounds over time by rewarding structures that work, dissolve, and are safe. These are what make the platforms of Insilico Medicine and Exscientia stand out. AlphaFold 3, which came out in 2024, takes things even further by working with DNA, RNA, and ligands in multi-modal complexes. This cuts the time it takes to find a structure from months to minutes.
These tools use omics data, such as genomics and proteomics, to find disease mechanisms that aren’t easy to see. Natural language processing, on the other hand, looks for new ways to use literature, just like BenevolentAI’s knowledge graphs do.
Benefits that matter
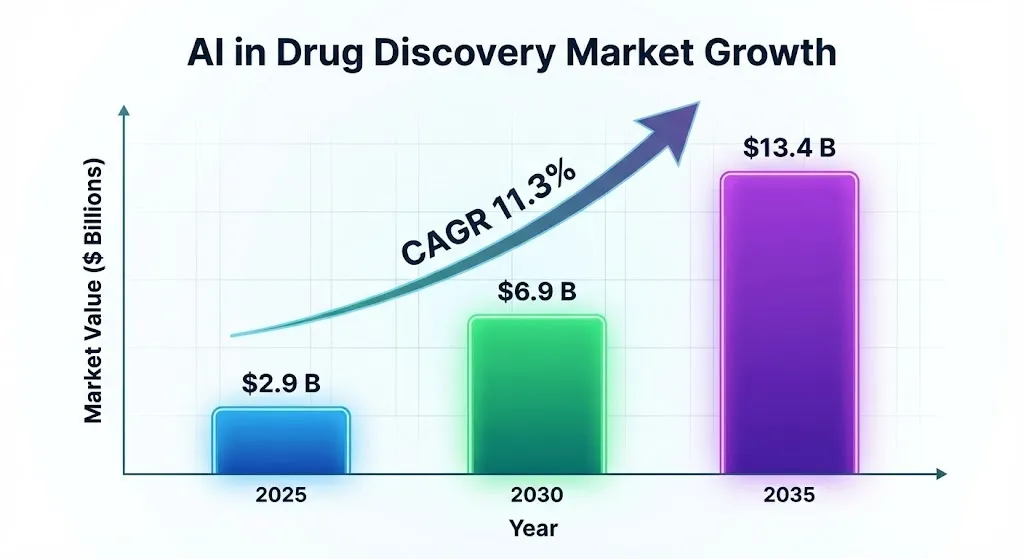
AI cuts the time it takes to make a drug by 30–40%, and the early discovery phases go from years to months. This could save each drug an average of $2.8 billion. Market predictions back this up: the AI drug discovery industry will grow from $2.9 billion in 2025 to $13.4 billion by 2035, with a compound annual growth rate (CAGR) of 11.3%.
The infographic below shows how AI can significantly reduce the time required at different stages of the drug discovery pipeline.
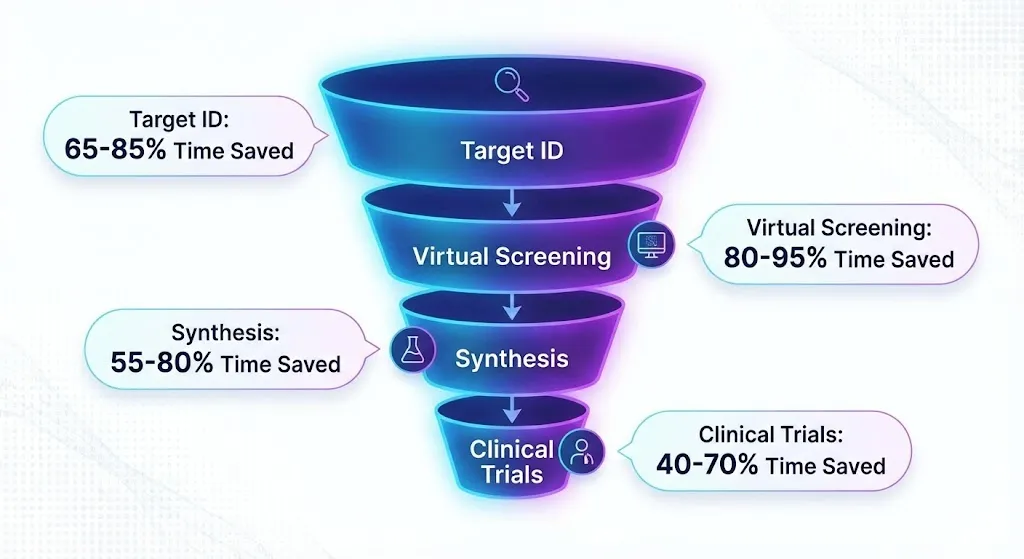
AI gets rid of candidates who aren’t useful before the lab, which makes it more likely that they will succeed and less likely that they will drop out, from 90% to less than 70% in the early stages. We believe that the market value will reach $16 billion by 2034 because there are so many chronic diseases and a need for research and development.
Here is a comparison table summarizing the key differences between traditional and AI-driven drug discovery.
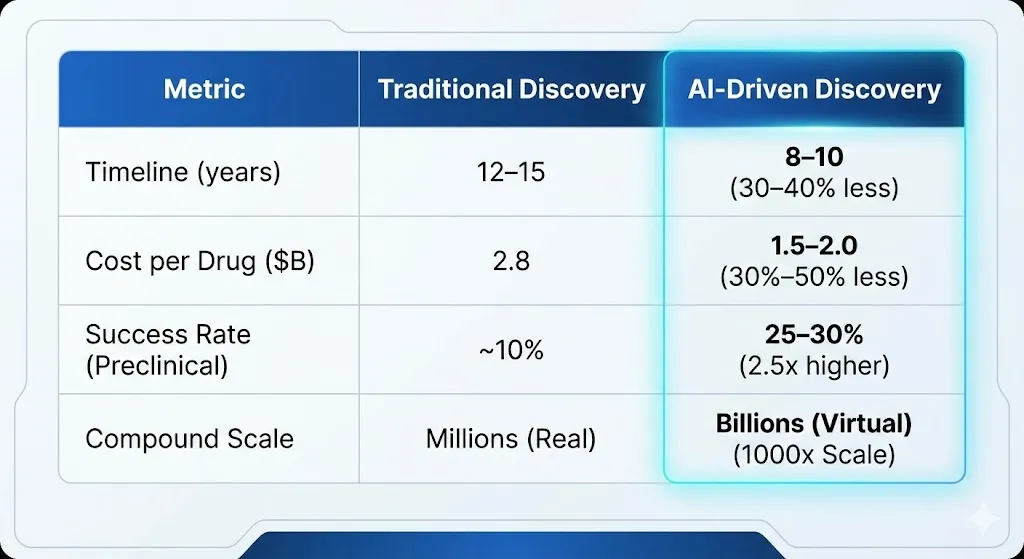
We tested millions of real compounds and billions of virtual compounds at a scale of 1000x.
Case Study: ISM001-055 from Insilico Medicine
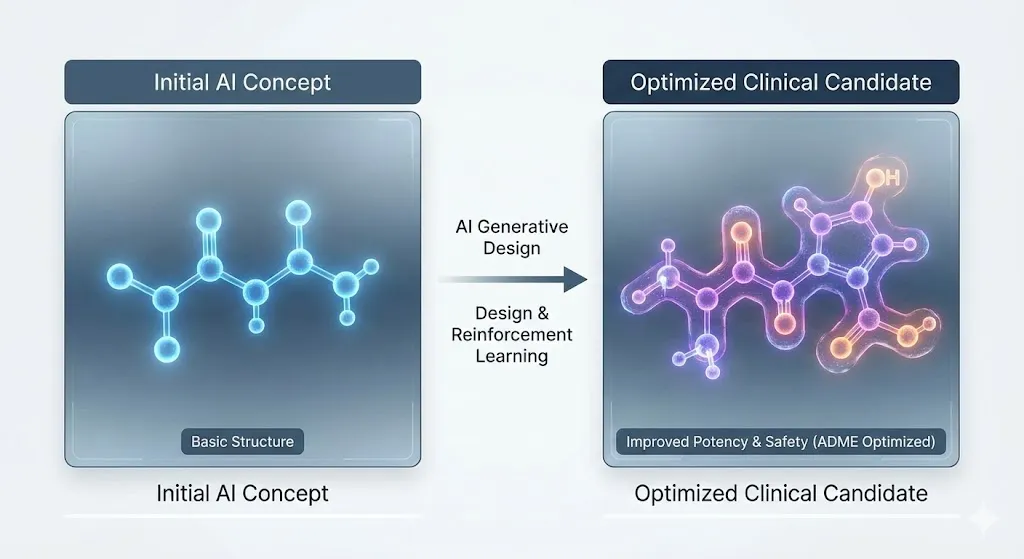
We look at Insilico Medicine’s successes with ISM001-055 (INS018_055), a generative AI-designed pan-fibrotic inhibitor for idiopathic pulmonary fibrosis (IPF). The platform produced 15 million virtual compounds, but only 60 of them were tested in a lab. It usually takes four to five years to go from finding a target to filing an IND, but this time it only took 18 months. As of 2026, a candidate that can get into the brain is now in Phase II trials.
This “moonshot” proves that end-to-end AI works: reinforcement learning made the molecule stronger and better at its ADME properties, which sped up a field that had been stuck for decades. The first data show that the treatments work well, which means they are the best in their class.
The following image illustrates how AI generative design and reinforcement learning optimize a molecule from a basic structure to a clinical candidate
A Case Study of Exscientia’s OCD Treatment
In 2020, Exscientia became the first company to make a drug that was designed by AI and tested on people with obsessive-compulsive disorder (OCD). They were partners with Sumitomo Pharma. They used deep learning to improve multiple targets on their platform, which helped them create the candidate in 12 months, which is 75% faster than the average in the industry.
By 2026, several Exscientia molecules will be undergoing Phase I/II clinical trials for cancer and immune system research. Automated chemistry will be right 75% of the time. This shows how good AI is at making exact plans for tough jobs.
Below is a summary table of key AI drug candidates and their milestones as of 2026.
Look at the Case Study: The Benefits of Combining Recursion and AlphaFold
Recursion Pharmaceuticals uses AI for phenotypic screening, which shows how cells respond to different drugs. This helps them find new places to look for cerebral cavernous malformation (CCM). Nvidia is helping them finish Phase II of their AI platform by 2026. It looks for diseases that are hard to find with normal methods.
Adding AlphaFold structures to predictions makes them more accurate, which leads to treatments that are six times more common than cystic fibrosis treatments.
The following image depicts a modern, high-tech pharmaceutical lab where AI and robotics are integrated into the discovery workflow.
What’s happening now and what’s going to happen next
More than 300 AI-biotech partnerships are doing well as of January 2026. Isomorphic Labs and BenevolentAI are teaming up with big drug companies like AstraZeneca, Pfizer, and Novartis. The most common type of AI is generative AI, and diffusion models make new molecules. NLP makes drug repurposing 40% more effective.
The market is growing by 29.7% every year, and trends are moving toward using edge AI and multi-omics integration to make predictions in real time.
The bar chart below shows the projected market growth of AI in drug discovery from 2025 to 2035.
Issues and Ways to Fix Them
Models don’t have enough information, and when they miss rare diseases, biases make things worse. We fight back by making fake data and using federated learning. It is still hard to explain things; regulators don’t trust black-box predictions. That’s why SHAP and other XAI methods are used to make outputs easier to understand.
AI needs to be able to last long enough to meet its computing needs, and ethical issues like intellectual property rights for AI-made molecules need strong frameworks.
What You Can Expect in the Future
We want AI to cut approval times in half and make personalized medicines by looking at patient genomes in real time with multimodal LLMs by 2030. Quantum-AI hybrids will be able to reach chemical spaces that are hard to get to and hit proteins like KRAS that can’t be drugged.
Precision oncology and neurodegeneration present numerous opportunities, with AI facilitating the development of fifty percent of all new pharmaceuticals. But groups that work across disciplines need to get rid of silos.
The Best Ways to Use AI
Hybrid Human-AI Workflows: Use domain knowledge to improve models for validation loops.
Different Datasets: Create multi-omics repositories to reduce bias.
Regulatory Alignment: Early use of the FDA’s XAI rules.
Scalable Infrastructure: Use cloud GPUs for DMTA cycles that happen over and over again.
Ethical Audits: Before using ethical audits, check for bias.
FAQ
How does AI help with the search for new drugs? In drug discovery, AI uses machine learning to guess how molecules will interact, create new compounds, and make leads better. This means that there is a lot less manual screening to do.
What are the advantages of using AI to search for new drugs? Some of the best things about it are that timelines are 30–40% shorter, costs are 30–50% lower, success rates are higher, and you can access huge virtual libraries.
Demonstrate the application of AI in drug discovery. Some examples are Insilico’s INS018_055 (Phase II, IPF), Exscientia’s OCD drug (clinical trials), and Recursion’s CCM therapy.
What problems does AI drug discovery face? New molecules have problems with data biases, model explainability, high computing needs, and following the rules.
What will AI do with drugs in the future? By 2030, people will get therapies in a different way because of drugs that are made just for them, quantum integrations, and faster development times.





Pingback: The Future of Personalised Medicine: AI and Your DNA - Bing Info